Water Quality and Cleaning Success

by Tom Forsythe & Michael Adams
The differences between hard and soft water have long been discussed for all sorts of applications. Hard water in the professional cleaning industry is known to play havoc with professional equipment.
As stated in their warranties, three major truckmount manufacturers will not cover malfunctions or component failures caused by hard water scale. Also, as you increase the heat in your equipment, your hard water scale issues will be accelerated.
Generally speaking, it is known that cleaning solutions do not perform as well with hard water compared to soft water. However, there have been no industry accepted criteria to put numbers to the performance differences. This article will attempt to provide clarity on the difference between hard and soft water for one product category: Extraction rinses.
Extraction rinses’ water testing
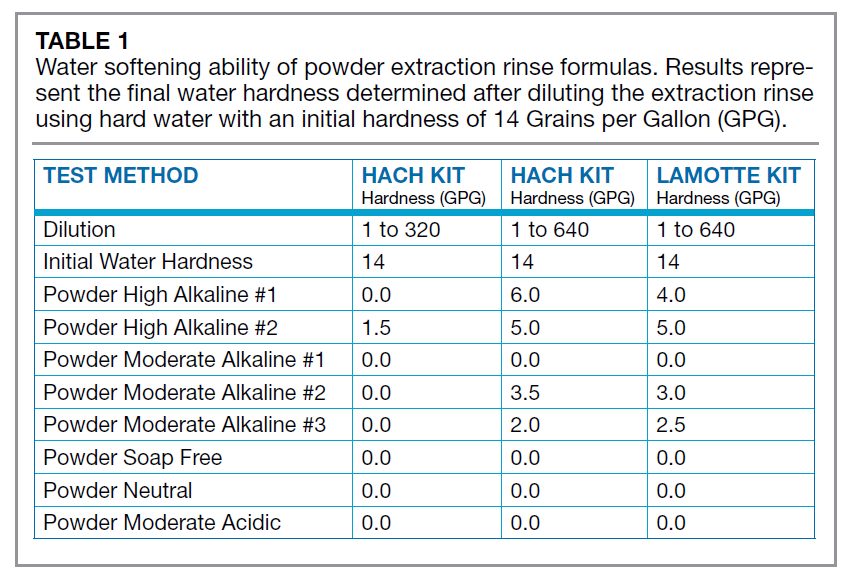
There are several test methods available in the industry to determine the hardness of water. During the authors’ evaluation, two test methods were used: The Hach Hardness Test Kit and LaMotte Total Hardness Test Kit. Results from the two test methods were compared to make sure there was no interference from the chemical components in any of the extraction rinse products. The two test methods provided similar results.
A total of 30 raw materials commonly used in rinse agents and their ability to soften hard water at 14 grains per gallon (GPG) were tested. Each ingredient was tested at 1 part raw material to 320 parts water by weight. Hardness was measured as quickly as possible since most equipment in the carpet cleaning industry uses a last step chemical injection system, meaning the cleaning solution is mixed with water for a limited amount of time. Water softening does not happen instantaneously. Generally, it was found that it took up to two minutes for the water to be totally softened.
Four primary ingredients in powdered formulations resulted in a water hardness of 0 to 8 GPG. Depending on the water hardness and dilution, it was identified in some cases that all of these ingredients could be used up in softening the water and not be available for cleaning. This static model allows for more time to soften the water, whereas the last step chemical injection system may not, depending upon how long the cleaning solution is in the line. One of these static tests indicated that approximately 85 percent of the product’s cleaning ingredients were used in water softening. In these situations, it is reasonable to assume this rinse would only clean a little better than water alone.
Next, 16 extraction rinse cleaning solutions, with 1 part formula to 320 parts water by weight, were tested. The liquid extraction rinses did little to the water’s hardness, removing at most 2 GPG from 14 GPG to 12 GPG. The powdered extraction rinses, in all instances but one, softened the water from 14 GPG to 0 GPG. This demonstrates the impact of powdered ingredients in terms of softening water. The table below illustrates these results.
We theorized that if we had the formula and the water softening ability for both the raw materials individually and the formulas collectively that we could come up with a percentage of the formulas used in softening water. All of the raw materials that softened the water contributed alkalinity to the cleaning system.
We also theorized that if the raw materials were used to soften water, then the alkalinity would also be used. This would mean that the pH of the diluted formulas would be different in soft and hard water at different dilutions. The greater the dilution, the more potential difference between the pH readings of the soft and hard water solutions should be realized. Even if there is not a difference in pH, that does not mean that there is not a difference in available alkalinity.
The alkaline and moderate alkaline formulas do not see much change in pH as there are other ingredients that do not soften water which provide alkalinity. The soap free, neutral and moderate acidic formulas have water softening agents with a higher pH than the pH of the total formula.When these water softening ingredients were bound up in softening water, the pH of the solutions dropped. We hypothesize that the water softening agents’ alkalinity was neutralized to some degree by being bound to the hard water, as seen in the table below.
In short, about 20 percent of ingredients in extraction rinses available for us to test are outstanding water softeners. However, in powdered extraction rinses, five ingredients form the backbone of these formulas. Many ingredients perform several functions, but those with water softening characteristics will be bound with hard water ions before coming in contact with the carpet. Static models show that up to 90 percent of some powdered formulas will be used to soften water at 14 GPG (45 percent at 7 GPG) and not be available for cleaning functions like providing alkalinity, soil suspension, enhancement of free rinsing, anti-redeposition, etc.
The truckmounts’ dynamic model, using last step chemical injection systems, will never reach the 90 percent water softening threshold since it takes about two minutes for water softening to be completed. When in action, water will move through the solution hoses at a pace fast enough (30 to 45 seconds for 250 feet of solution hose) to prevent total water softening.
Formulators are not adding these ingredients to soften the water, but to accomplish other purposes. Also, water softening salt is $.15 per pound, while most powdered extraction rinses are at least $6 per pound. Allowing your powdered rinse to be used as a water softener is expensive, and to make up for the loss of cleaning power, you would need to reduce your dilution from 1 to 640 to 1 to 320. It does not take too long at this level of waste to buy your own water softener.
Make the smart move
It is said that wisdom is the application of knowledge. Congratulations to those who use water softeners, as you are protecting your equipment and also providing better cleaning. In most cases, you have learned from both knowledge and experience and have made appropriate adjustments to your cleaning system.
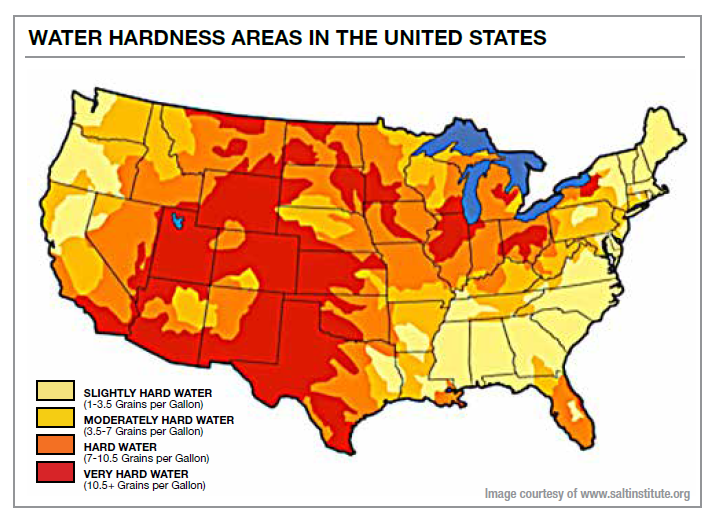
For those of you who are still using hard water, there are a few options. The first step is to determine the GPG of the water in your area. In some cases it may be useful to test the water you are using. What is your number? Determine the hardness of water where you live. The higher the number the more important this article is to your cleaning operation. A high-heat truckmount could move you from hard water to very hard water in terms of the amount of scale that impacts your rinse and equipment.
The easiest step is to buy and properly maintain a water softener as suggested by truckmount manufacturers if your hard water is above 3.5 GPG. This reduces your overall costs,
such as chemical usage and maintenance costs.
Another option is to use liquid extraction rinses, which contain less ingredients used softening the water. Their performance is also impacted, but not to the degree of powdered extraction rinses.
Or, you can add a scoop of soap free rinse (no fragrances at a pH of 10) to your pre-spray, which distributes the rinse agents through the carpet in the pre-treatment phase. During the extraction process, the hard water will come in contact with the emulsified soil surrounded by the rinse agents, improving the soil removal. These rinse agents would not be used up in softening the hard water, allowing their full cleaning functionality to be utilized.
A last option is to continue to do as you’ve always been doing. Someone of this opinion would probably never read this article. Often, you hear some say they have 30 years of experience, but often this means they have one year of experience repeated 30 times. Let’s hope we truly gain a year of experience.

Tom Forsythe

Michael Adams
Tom Forsythe has worked as a chemist for Bridgepoint Systems for 16 years and has developed more than 175 products. He has made 16 rinse products over the years, 10 liquid and six powdered formulations.
Michael Adams joined Bridgewater in 2015 and serves as the EHS manager/compliance manager for the company. He has worked in the chemical products industry for the last nine years and has a Master’s of Science in Chemical Engineering from the University of Utah.